KPV (Lys–Pro–Val) is the C‑terminal tripeptide fragment of the α‑melanocyte‑stimulating hormone (α‑MSH). As a small bioactive peptide, KPV reproduces many of the anti‑inflammatory, antimicrobial, and tissue‑protective activities of α‑MSH, while avoiding full melanocortin‑receptor signaling in some contexts
R$635,00
10 in stock
| From (units) | Packing multiple | Unit price |
|---|---|---|
| 30 | multiples of 10 | R$564,00 |
| 500 | multiples of 10 | R$494,00 |
| 3.000 | multiples of 10 | R$423,00 |

KPV (Lys–Pro–Val) corresponds to the final three amino acids of α‑melanocyte‑stimulating hormone (α‑MSH), a peptide derived from proopiomelanocortin (POMC). As a compact, bioactive peptide, KPV retains key anti‑inflammatory, antimicrobial, and tissue‑protective properties of the parent hormone while displaying a more selective signaling profile, without triggering the full spectrum of melanocortin‑receptor activation.
In research settings, KPV has been investigated for the following actions:
The KPV sequence serves as a useful research tool for studying innate immune regulation, mucosal defense, and novel anti‑inflammatory mechanisms of short peptides.
| Property | Value |
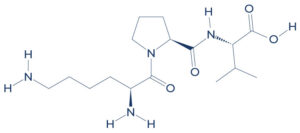
| Chemical formula | C₁₆H₃₀N₄O₄ |
| Molecular weight | 342.43 g/mol |
| Synonyms | α‑MSH (11‑13), ACTH (11‑13), MSH (11‑13) |
Source: Científico
KPV is supplied as a lyophilized powder. Lyophilization enhances stability and shelf life while preserving purity and molecular structure during storage. No fillers or bulking agents are used in this process.
Biopelabs advises: this material is provided exclusively as a chemical reagent for research purposes. Its use is restricted to in vitro assays and experimental work in a controlled laboratory setting. The information provided is strictly educational and informational. Handling must be performed only by qualified professionals. This product is not classified as a medicine, food, or cosmetic and must not be used, marketed, or described as such.
KPV is a tripeptide derived from the C‑terminal region of α‑MSH (alpha‑melanocyte‑stimulating hormone) and has attracted growing interest in the scientific literature for its anti‑inflammatory, immunomodulatory, and antimicrobial activity. Preclinical studies indicate that, even in its short form, KPV reproduces several biological properties of full‑length α‑MSH, making it a relevant compound for experimental investigations.
Research shows that KPV and other C‑terminal α‑MSH fragments can modulate inflammatory responses through multiple pathways. Described mechanisms include reduced release of pro‑inflammatory cytokines, increased intracellular cAMP levels, and inhibition of the NF‑κB signaling pathway, which plays a central role in inflammation.
In vitro and in vivo assays suggest that these small melanocortin fragments exert not only immunomodulatory effects but also direct antimicrobial activity, expanding their potential use in studies of inflammation and host defense.
In preclinical models, KPV and its derivatives, such as dimeric forms, have shown the ability to reduce production of inflammatory mediators, including TNF‑α and nitric oxide, in responses triggered by lipopolysaccharides (LPS).
These effects have been observed both in human peripheral blood mononuclear cells and in animal models exposed to endotoxins, indicating a significant attenuation of systemic inflammatory responses. Such findings support the interest in KPV as a research target in experimental contexts related to endotoxemia and sepsis.
Beyond modulating inflammation, studies report that KPV has direct antimicrobial activity against various microorganisms. Experimental evidence documents its effects against pathogens such as Candida albicans, Escherichia coli, and Staphylococcus aureus.
This combination of effects—control of inflammation and antimicrobial action—suggests that KPV may contribute to pathogen clearance while simultaneously reducing inflammation‑induced tissue damage, making it an attractive compound for research into infection and immunity.
In the field of tissue repair, KPV has been studied for its ability to accelerate healing of epithelial tissues, including the cornea. Experimental models show that the peptide promotes closure of corneal epithelial wounds, an effect that appears to be partially mediated by nitric‑oxide signaling.
These results indicate that KPV may be useful in research focused on regeneration of mucosal and epithelial surfaces, particularly in sensitive tissues that require tight control of local inflammation.
An important aspect of KPV is that some of its actions appear to occur independently of classical melanocortin receptors such as MC1R. Mechanistic studies suggest that the peptide may interact directly with cell membranes, intracellular targets, or alternative receptors.
This non‑conventional mode of action increases scientific interest in KPV, as it points to mechanisms distinct from those of other melanocortin‑family peptides.
More recent work places KPV and related α‑MSH fragments within a broader therapeutic context, including potential antifibrotic, anti‑inflammatory, and tissue‑protective effects in different organ systems.
These findings broaden the research scope of KPV and encourage exploratory studies on its role in fibrosis and tissue‑remodeling processes.
Because it is a short peptide, KPV can undergo rapid degradation and clearance in the body. Consequently, ongoing research focuses on developing KPV derivatives, such as dimeric forms or lipid‑modified analogues, as well as delivery systems that enhance its stability and tissue persistence.
These strategies aim to improve local bioavailability, experimental efficacy, and the therapeutic index of the peptide in preclinical studies.
Singh, M., & Mukhopadhyay, K. (2014). Alpha‑melanocyte‑stimulating hormone: An emerging anti‑inflammatory and antimicrobial peptide. Biomedical Research International, 2014, 874610. Summary review of α‑MSH and C‑terminal fragments (including KPV) as antimicrobial and anti‑inflammatory agents. https://pmc.ncbi.nlm.nih.gov/articles/PMC4130143/
Star, R. A., et al. Classic review on α‑MSH and its anti‑inflammatory potential, covering mechanisms of α‑MSH fragments and anti‑inflammatory signaling. https://pmc.ncbi.nlm.nih.gov/articles/PMC2095288/
Gatti, S., Carlin, A., Sordi, A., et al. (2006). Inhibitory effects of the (CKPV)₂ peptide on endotoxin‑induced host reactions. Journal of Surgical Research, 131(2), 209–214. Demonstrates that (CKPV)₂ and KPV reduce LPS‑induced TNF‑α and NO in vitro and in vivo. https://pubmed.ncbi.nlm.nih.gov/16413580/
Singh, M., & Mukhopadhyay, K. (same review as above) — discusses direct antimicrobial activity of C‑terminal α‑MSH peptides (KPV), including candidacidal and antibacterial data. https://pmc.ncbi.nlm.nih.gov/articles/PMC4130143/
Kozub, J. A., et al. COOH‑terminal tripeptide α‑MSH(11–13) effects on corneal epithelial wound healing: role of nitric oxide. Corneal wound‑healing study demonstrating KPV/α‑MSH(11–13) effects. https://pubmed.ncbi.nlm.nih.gov/16965771/
Mechanistic evidence — MC1R independence: data showing that KPV‑like peptides do not always act via classical MC1R binding (representative mechanistic figures and analysis). https://www.researchgate.net/figure/KDPT-does-not-act-via-a-MSH-binding-sites-does-not-bind-to-MC-1R-and-has-no_fig3_45101538
Dinparastisaleh, R., & Mirsaeidi, M. (2021). Antifibrotic and anti‑inflammatory actions of alpha‑melanocyte‑stimulating hormone: New roles for an old player. Pharmaceuticals, 14(1), 45. Review covering antifibrotic and anti‑inflammatory roles of melanocortin peptides and fragments. https://www.mdpi.com/1424-8247/14/1/45
Mechanistic cAMP/NF‑κB signaling studies on α‑MSH‑derived peptides, detailing modulation of cAMP/NF‑κB pathways and additional mechanistic data on short fragments. https://www.sciencedirect.com/science/article/pii/S0022202X15307697
Recent review on KPV peptide delivery and translational perspectives, summarizing advances in formulation and delivery strategies for KPV. https://www.scitechnol.com/peer-review/recent-advances-in-kpv-peptidedelivery-8OYD.php?article_id=18216

The content was reviewed by Dr. Ky H. Le, MD. Dr. Ky H. Le is a family physician in Aiea, Hawaii. He received his medical degree from St. George’s University School of Medicine and has been practicing for over 20 years. He has expertise in the treatment of obesity, diabetes, hypertension, and high blood pressure, among other conditions—see all areas of specialization at https://health.usnews.com/doctors/ky-le-371599#expertise. Dr. Ky H. Le accepts Medicare, Aetna, Humana, Blue Cross, and United Healthcare.
Reference: https://health.usnews.com/doctors/ky-le-371599#expertise

Only logged in customers who have purchased this product may leave a review.


Reviews
There are no reviews yet.