MOTS-c is a mitochondria-derived peptide composed of 16 amino acids, encoded by the 12S rRNA region of the mitochondrial genome. It plays significant roles in metabolic homeostasis, nuclear gene expression, and has potential therapeutic applications for metabolic dysfunction, insulin sensitivity, and age-related diseases
R$1.168,00
Out of stock
| From (units) | Packing multiple | Unit price |
|---|---|---|
| 30 | multiples of 10 | R$1.038,00 |
| 500 | multiples of 10 | R$910,00 |
| 3.000 | multiples of 10 | R$780,00 |

MOTS-c (Mitochondrial Open Reading Frame of the 12S rRNA-c) is a mitochondria-derived peptide discovered in 2015 that functions as a mitochondrially encoded regulatory signal. This 16-amino-acid peptide is encoded within the mitochondrial genome in the 12S rRNA gene and represents an important link between mitochondrial function and metabolism.
MOTS-c primarily acts in skeletal muscle and functions as a metabolic regulator by activating the AMPK (AMP-activated protein kinase) pathway, which is crucial for cellular energy homeostasis
| Properties | Value |
| Molecular Formula | C101H152N28O22S2 |
| Molecular Weight | 2174.6 g/mol |
| Synonyms: | 1627580-64-6, UNII-A5CV6JFB78, MOTS-c (human) (trifluoroacetate salt), A5CV6JFB78 |
Main Structure of the Peptide Mots_c
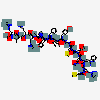
Source: PubChem
The peptides undergo a lyophilization process, a technique that contributes to greater stability and durability while preserving purity and molecular structure during storage. It is important to note that no fillers are used during this process.
Biopelabs advises: this material is provided exclusively as a chemical input for research purposes. Its use is restricted to in vitro assays and experimental activities in laboratory settings. The information presented is strictly for informational and educational purposes. Handling must be carried out only by properly qualified professionals. The product is not classified as a medication, food, or cosmetic, and must not be used, marketed, or described as such
MOTS-c is a recently discovered mitochondrial peptide that has generated significant interest in the scientific community due to its potential role in metabolic regulation, maintenance of muscle function, neuroprotection, and processes associated with healthy aging. Although studies are still largely in experimental stages, results from preclinical models point to a broad field of investigation for metabolic disorders and age-related conditions.
Under conditions of metabolic stress, MOTS-c is capable of translocating to the cell nucleus, where it directly influences nuclear gene expression. This process occurs in a manner dependent on the activation of AMPK, a central enzyme in cellular energy balance regulation.
During this nuclear regulation, MOTS-c interacts with stress-responsive transcription factors such as NRF2, modulating genes involved in antioxidant response, glucose metabolism, and cellular energy adaptation.
Studies demonstrate that activation of the AMPK pathway by MOTS-c contributes to improved insulin sensitivity and increased energy expenditure, particularly in contexts of metabolic dysfunction. In experimental models, this activation has been shown to be relevant in preventing metabolic alterations associated with hormonal deficiency.
Additionally, MOTS-c has demonstrated protective effects against diet-induced obesity and insulin resistance, promoting increased glucose utilization in skeletal muscle. This effect is associated with inhibition of the folate cycle, which favors AMPK activation and maintenance of metabolic homeostasis.
In models simulating the postmenopausal period, administration of MOTS-c resulted in reduced fat mass, decreased inflammatory markers, and preservation of brown adipose tissue activity, preventing weight gain and impairments in insulin response.
MOTS-c has also been studied for its neuroprotective properties, particularly due to its anti-inflammatory action in the central nervous system. Research indicates that the peptide reduces activation of astrocytes and microglia, leading to decreased production of pro-inflammatory cytokines associated with neuroinflammation.
This reduction in the cerebral inflammatory environment has been correlated with improvements in cognitive function and memory in experimental models of neurodegenerative diseases and traumatic brain injury.
Beyond inflammatory control, MOTS-c contributes to metabolic support in the brain by stimulating processes such as lipid oxidation, which provide additional energy to neural tissue following injury. This energy support helps preserve neuronal function and reduce cellular damage.
Experimental studies have also demonstrated improvements in recognition and spatial memory, suggesting that MOTS-c may positively influence memory formation and consolidation under conditions of cognitive impairment.
Despite these promising findings, further research is required to fully understand the mechanisms of action of MOTS-c in the brain and to optimize its administration strategies in clinical contexts.
In skeletal muscle tissue, MOTS-c plays an important role in regulating metabolism and proteostasis, influencing nuclear genes associated with the maintenance of muscle mass and function. It also helps myoblasts adapt to metabolic stress, a key factor in preserving muscle strength over time.
In animal models, treatment with MOTS-c resulted in significant improvements in physical performance across different life stages, including young, adult, and aged subjects. These findings suggest a potential protective effect against age-related physical decline.
Another relevant observation is MOTS-c’s ability to reduce levels of myostatin, a protein that inhibits muscle growth. This effect occurs through modulation of the PTEN/AKT/FOXO1 pathway, which is essential for balancing muscle protein degradation and synthesis.
In humans, physical exercise has been shown to increase MOTS-c expression in skeletal muscle and circulation, reinforcing its role as an exercise-induced regulator of physical capacity and metabolic health.
These data position MOTS-c as a compound of interest in research focused on sarcopenia, insulin resistance-associated muscle atrophy, and other conditions involving loss of muscle mass and function.
The body of experimental evidence suggests that MOTS-c may play a relevant role in strategies aimed at promoting longevity and healthy aging. Its ability to modulate metabolism, preserve muscle function, improve physical performance, and support neural protection makes it a promising target for future research in anti-aging therapies.
Additionally, genetic studies have identified a specific polymorphism in the MOTS-c coding region associated with exceptional longevity in certain populations, indicating a possible genetic basis for its aging-related effects.
Kim, K., et al. (2018). The mitochondrial-encoded peptide MOTS-c translocates to the nucleus to regulate nuclear gene expression in response to metabolic stress. Cell Metabolism, 28(3), 516–524.e7. https://doi.org/10.1016/j.cmet.2018.06.008
Lu, H., et al. (2019). MOTS-c regulates adipose homeostasis to prevent ovariectomy-induced metabolic dysfunction. Journal of Molecular Medicine, 97, 473–485. https://doi.org/10.1007/s00109-018-01738-w
Lee, C., et al. (2015). The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metabolism, 21(3), 443–454. https://doi.org/10.1016/j.cmet.2015.02.009
Li, F., et al. (2024). Neuroprotective mechanism of MOTS-c in TBI mice. Drug Design, Development and Therapy, 18, 2971–2987. https://doi.org/10.2147/DDDT.S460265
Jiang, J., et al. (2021). Peripheral administration of a cell-penetrating MOTS-c analog improves memory and attenuates impairment. ACS Chemical Neuroscience. https://doi.org/10.1021/acschemneuro.0c00782
Reynolds, J., et al. (2021). MOTS-c is an exercise-induced mitochondrial regulator of age-dependent physical decline. Nature Communications. https://doi.org/10.1038/s41467-020-20790-0
Kumagai, H., et al. (2021). MOTS-c reduces myostatin signaling and muscle atrophy. American Journal of Physiology. https://doi.org/10.1152/ajpendo.00275.2020
Mohtashami, Z., et al. (2022). MOTS-c in human aging and age-related diseases. International Journal of Molecular Sciences, 23. https://doi.org/10.3390/ijms231911991
Fuku, N., et al. (2015). MOTS-c: a player in exceptional longevity. Aging Cell, 14, 921–923. https://doi.org/10.1111/acel.12389

This content was reviewed by Dr. Ky H. Le, MD. Dr. Ky H. Le is a family physician based in Aiea, Hawaii. He earned his medical degree from St. George's University School of Medicine and has been practicing for over 20 years. He has expertise in the treatment of obesity, diabetes, hypertension, and high blood pressure, among other conditions. Dr. Ky H. Le accepts Medicare, Aetna, Humana, Blue Cross, and United Healthcare.
Ref.: https://health.usnews.com/doctors/ky-le-371599#expertise

Only logged in customers who have purchased this product may leave a review.


Reviews
There are no reviews yet.